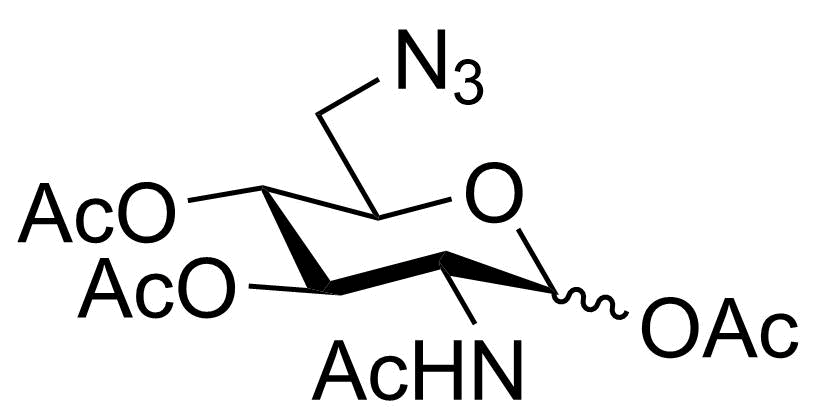
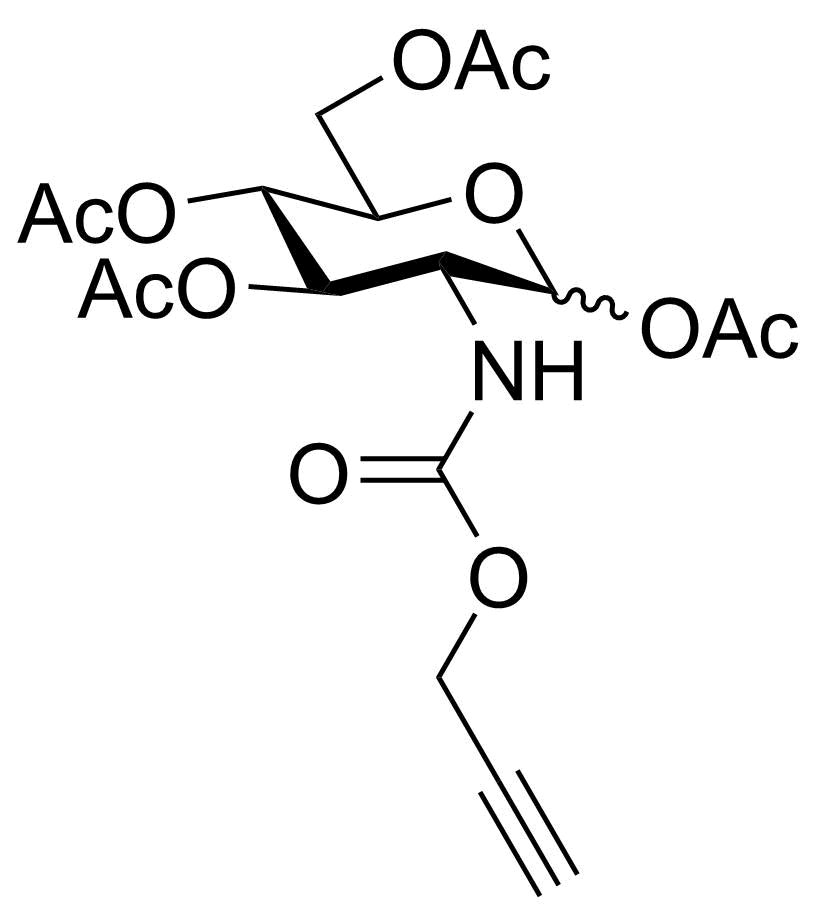
P-Neu5NAz
Metabolic label of sialylation
| Product ID: | SV2443 |
|---|---|
| Synonyms: | Ac5SiaNAz; Ac5Neu5NAz |
| Tags: | Azide, Bioorthogonal chemistry, Metabolic label, Neuraminic acid, Neuraminidase, Sialic acid, Sialidase |
| Product | Price | Estimated Shipping Time | Purchase |
|---|---|---|---|
|
|
€250.00 | 1-3 days | |
|
|
€425.00 | 1-3 days | |
|
|
€1,100.00 | 1-3 days |
Product information
-
Function
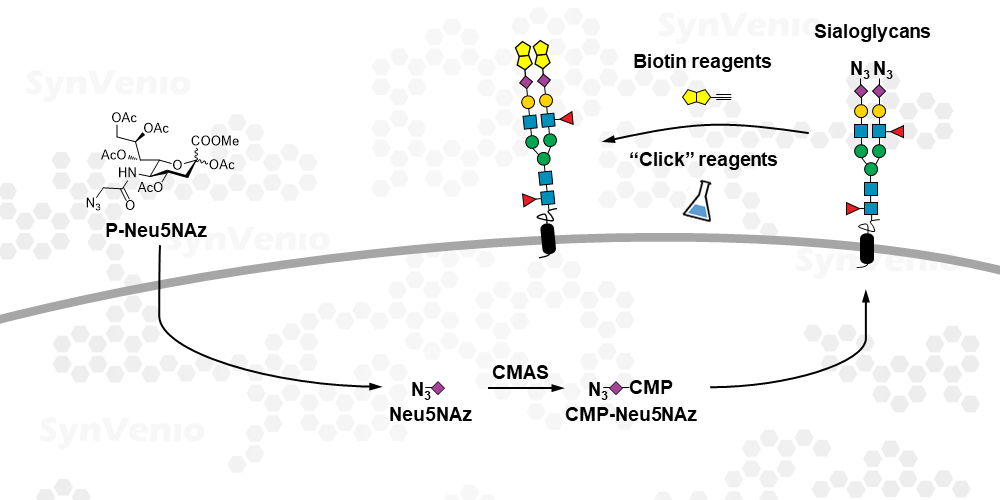
Unnatural sialic acid analog for metabolic labeling. Contains a Azide group for “click” chemistry. P-Neu5NAz is passively taken-up by cells and incorporated in cell surface sialoglycans. Alkyne reagents can be reacted with the Azide group on live and fixed cells.1 These azides can be modified with an affinity tag or fluorophore for example. Small molecules can also be attached to enhance the binding to Siglec receptors.2 In contrast to P-Neu5NPoc, P-Neu5NAz residues can be cleaved by sialidases and hence minimally perturb the turnover of Neuraminic acids.3 This has been studied in the context of viral infection. In this study, the use of P-Neu5NAz in host cells did to lead to a decreased influenza infection by blocking its neuraminidase whereas use of P-Neu5NPoc did.
-
Mode of action
P-Neu5NAz is passively taken-up by mammalian cells and converted into its CMP-SiaPoc derivative. This is used as a donor substrate for sialyltransferase enzymes and transferred to O-, N-glycans and glycolipids. P-Neu5NAz is a much more efficient metabolic label of sialic acid than ManNAc based derivative and also less toxic.1, 4
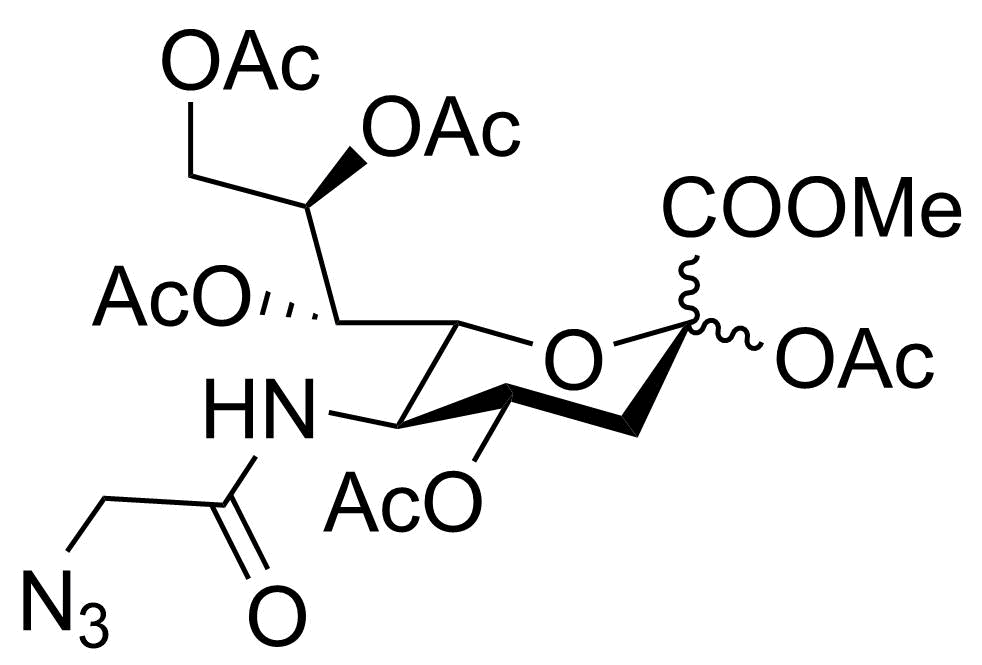
-
Applications
Metabolic labeling of sialoglycans for isolation, visualization and to improve Siglec binding
-
Handling
P-SiaNAz is soluble in DMSO and PBS and is added to cell culture from a stock solution. Incubation of the cells with P-SiaNAz for 3 days in needed to achieve the maximal metabolic incorporation. Three days is needed to achieve the turnover of sialylated glycans to achieve maximal metabolic incorporation.
-
Chemical Information
CAS No.: NA
SMILES: O=C(CN=[N+]=[N-])N[C@H]1[C@H]([C@H](OC(C)=O)[C@H](OC(C)=O)COC(C)=O)OC(C(OC)=O)(OC(C)=O)C[C@@H]1OC(C)=O
Chemical formula: C22H30N4O14
Molecular weight: 574.18
Purity: > 95%
Identity: 1H NMR
Shipping temperature: 20°C
Storage temperature: 0°C
Recommended Products
| Name |
|---|
| P-GlcNAc6Az |
| Fuc2F |
| XylNap |
| Neu5Ac3F |
| P-GlcNPoc |
Calculator
Dissolve the required mass in your desired stock volume.
Dilute the required volume of your stock solution to the desired final volume.